In the food processing, pharmaceutical production and cold chain logistics industries, accurate temperature and humidity monitoring with compliant data recording is critical to product safety, and FDA 21 CFR Part 11 is the global benchmark for data integrity— a must for regulatory compliance and global market access. Freshliance’s Tagplus-T/TH FDA temperature data loggers deliver a fully compliant solution, combining hardware adherence to FDA standards with a dedicated FDA software suite, redefining reliable, hassle-free environmental monitoring for high-regulation sectors.

The FDA temperature data logger monitors data reliably and store raw data in an encrypted read-only format, preventing manual alteration or deletion to ensure uncompromised data integrity. Connecting to a computer via the integrated USB design, it automatically generates encrypted, tamper-proof reports with summary data, trend charts and statistics. The temporary report mode enables mid-recording data retrieval without interrupting monitoring. Each device is factory-calibrated against reference instruments certified by CNAS/ISO17025 labs, ensuring data is NIST-traceable for unrivaled accuracy.
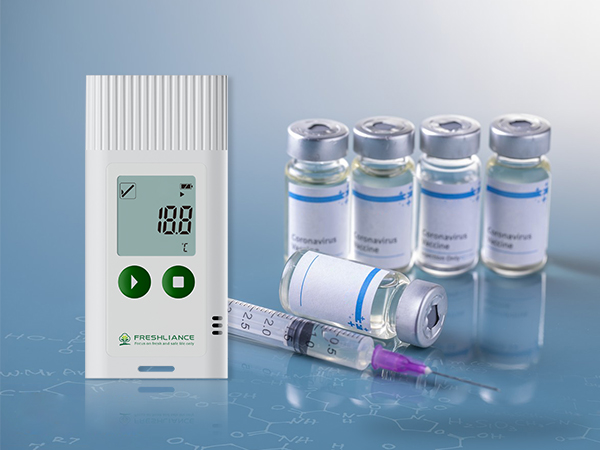
For 21 CFR Part 11 compliance, the software Freshliance Tools-FDA is built exclusively for strict audit trails, e-signatures and access control. It auto-tracks all critical operations, offers role-based access for admins, operators and auditors, binds e-signatures to key approvals, and provides an audit-grade central data platform with encryption.
Tagplus-T/TH PDF temperature data logger’s hardware-software integration meets all monitoring needs, from daily tracking to high-level regulatory audits. The FDA temperature data logger delivers accurate, traceable and secure data. Compliant to FDA CFR21, EN12830, CE and RoHS, it meets international food and pharma norms.

 English
English Español
Español Русский
Русский Français
Français Deutsch
Deutsch عربي
عربي 中文
中文



